VIDO-InterVac joins swine fever fight
U of S centre to work with ASF virus in Saskatoon
By GFM Staff
| 2 min read
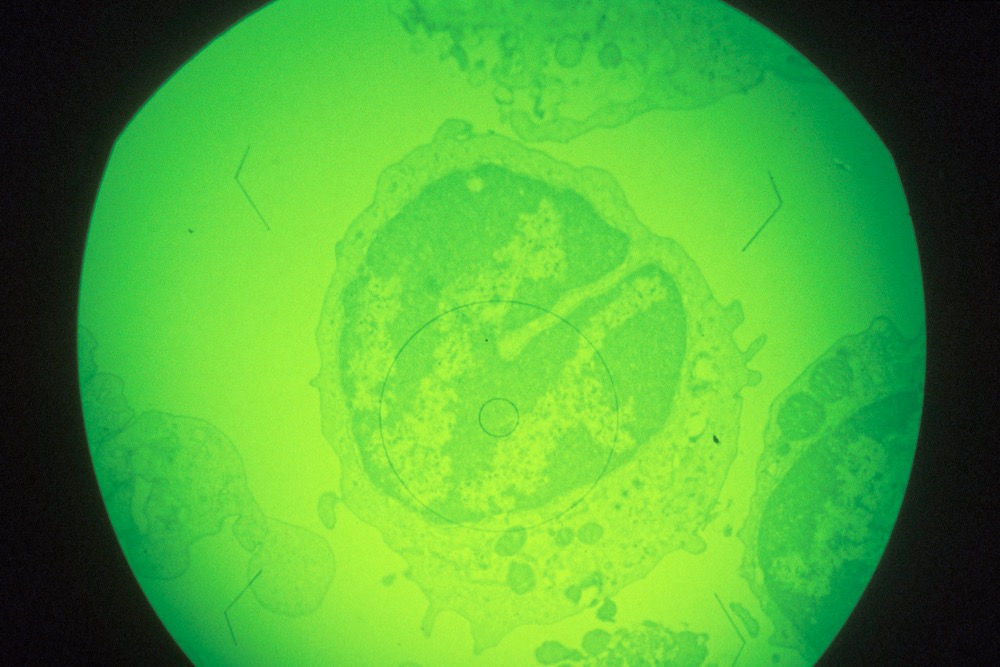
A macrophage (immune response) cell in early stages of infection with African swine fever virus, magnified about 1,000x. (Keith Weller photo courtesy ARS/USDA)
The University of Saskatchewan’s vaccine research and development arm is set to become Canada’s first non-government agency to work with the African swine fever virus.
Saskatoon-based Vaccine and Infectious Disease Organization-International Vaccine Centre (VIDO-InterVac) will start work with the virus this month at its Containment Level 3-Agriculture (CL3-Ag) facility, the Canadian Food Inspection Agency said in a release Wednesday.
VIDO-InterVac said it will work with researchers at the University of Alberta, CFIA and the South African Research Council to evaluate a “novel proprietary adenoviral vector vaccine” against the disease.
Adenoviral vectors are used as vaccine delivery systems against infectious diseases, carrying foreign genes to spur a specific response from antibodies or T-cells.
CFIA, in its release, described the planned research as “an important step towards the development and testing of vaccines and antivirals for ASF.”
ASF leads to haemorrhagic fever and death in infected domestic and wild pigs and, while it does not pose a food safety risk, has been responsible for “serious production and economic losses” in the hog sectors of several countries.
Unlike “classical” swine fever, there’s no approved vaccine or treatment yet available against ASF.
The ASF virus can be spread by live or dead pigs and pork products and/or via contaminated feed and objects such as shoes, clothes, vehicles, knives and equipment, due to its “high environmental resistance,” according to the World Organization for Animal Health (OIE).
As of the week ending Jan. 3, the OIE booked 23 countries as reporting new or ongoing outbreaks in farmed swine and/or wild hogs.
ASF’s effects on China in particular — where the hog herd has declined by over 40 per cent in the past year through infections and culls to control the disease — have roiled commodity markets. The virus has never been detected in Canada or the U.S.
“CFIA’s support increases Canada’s international contribution to combat the spread of ASF,” VIDO-InterVac director Dr. Volker Gerdts said in CFIA’s release.
“This is a prime example of how this CL3-Ag infrastructure supports national priorities against emerging infectious disease and the development of solutions that mitigate their impact.” — Glacier FarmMedia Network